This unit aims to cover the general building blocks of biology. Since this material will show up through all the other units, it is important to truly understand why these reactions/processes happen rather than purely memorizing them. Specifically, understanding how a molecule’s shape and bonding determines its function. If you are not aware how the chemistry of these molecules work, we highly recommend you watch this chemistry video to gain a quick understanding of how these molecules interact.
Monomers / Polymers
Monomers : Small, single molecules that act like building blocks
Polymers: Large molecules made by linking many monomers together in a chain.
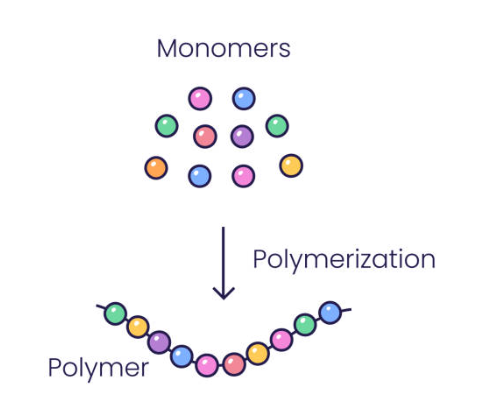
Monomers join together with a covalent bond to become a polymer chain.
| Monomers | Polymers |
| Monosacciride | carbodydrate |
| Amino Acid | protien |
| Nucleotide | nucleic acids |
| *fatty Acid | lipids |
NOTE: Memorize both the monomer and polymer names for your Unit 1 test, but going forward, most questions will reference the polymer form – lone monomers rarely exist alone in the body.
Dehydration Synthesis
Goal: Synthesize polymers from monomers
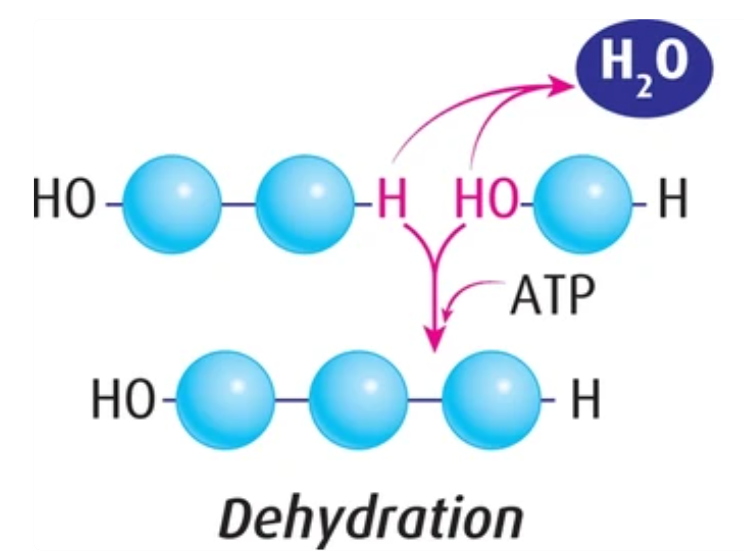
- Produces Water (by product)
- Requires energy (ATP)
- Low potential energy –> high potential energy
- Stores Energy (in polymers)
Hydrolysis
Goal: Break apart polymers to produce monomers
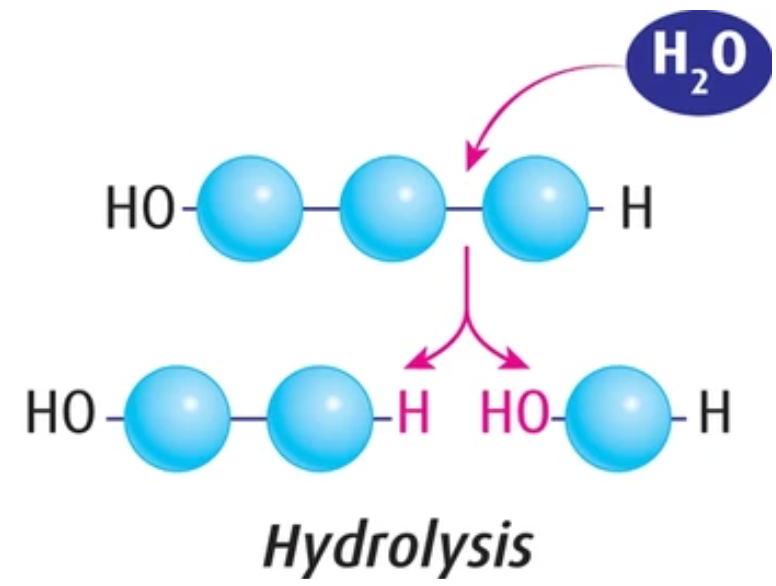
- Requires water
- Produces ATP
- High potential energy –> low potential energy
- Releases energy (breaks apart polymers into monomers)
Tips:
- This unit is all about how the structure of the molecule impacts its function and role
- Understanding the key differences of each type of molecule and connecting it with how that impacts its function (including examples) will help during tests