Definition and Function
- Non-polar
- Hydrophobic molecules
- Formula ratio: CHO
Structure and Polarity
- Fatty Acid –> long hydrocarbon chain (non-polar) + carboxyl group (polar, acidic)
- Amphiatic = has polar and non-polar
- Lipids – non-polar overall because of long carbon chain
4 Types of Lipids
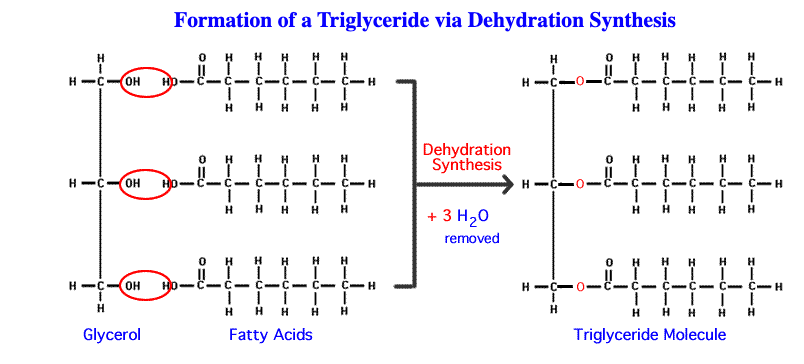
1. Tricglycerides
- Forms through dehrdration synthesis
- Made of a glycerol + 3 fatty acids
- Ester bonds join glycerol+fatty acids
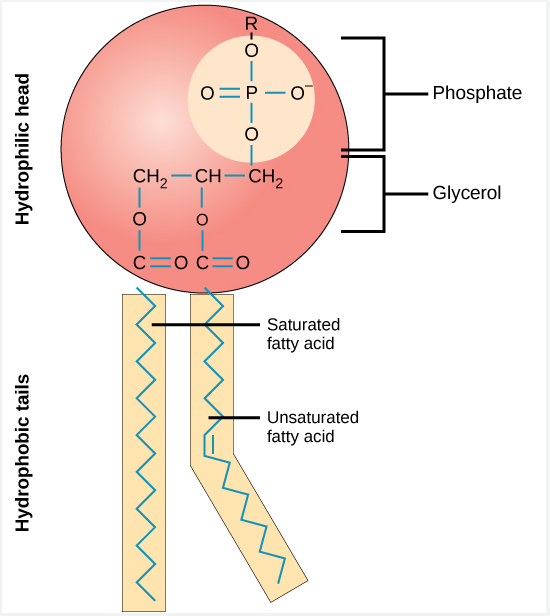
2. Phospholipids
- Glycerol + 2 fatty acids + phosphate head
- Polar head
- Non polar tail
- Forms Cell Memrbanes
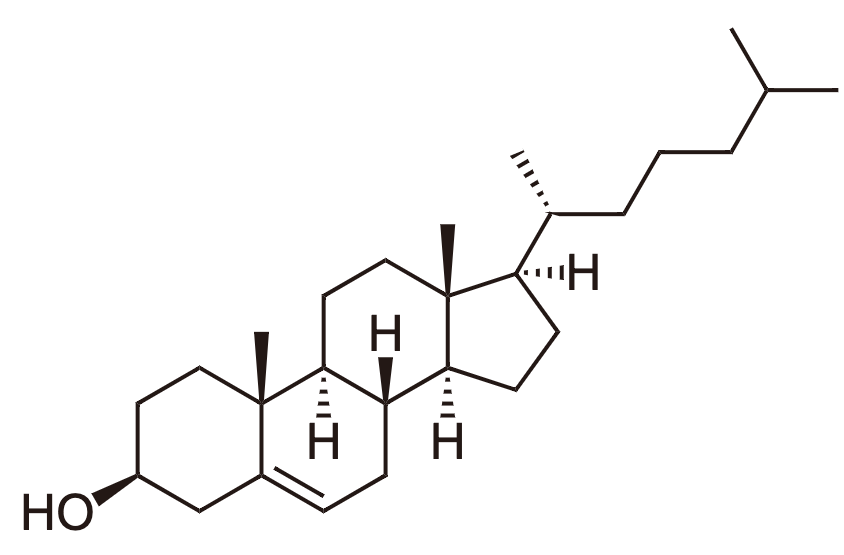
3. Steroids
- 4 fused carbon rings
- Cholesterol
- Estrogen
- Testorone
- Non-Polar / Hydrophillic
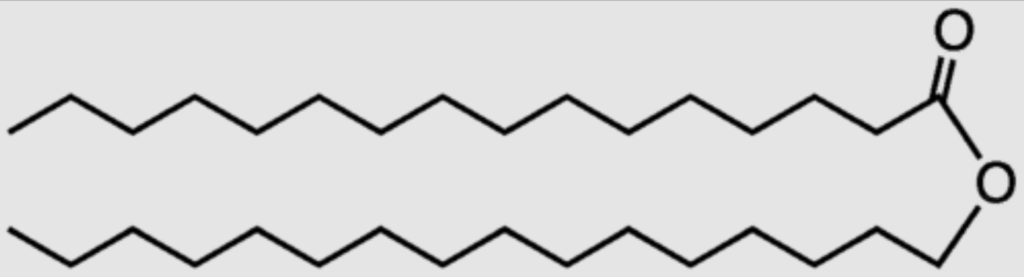
4. Wax
- Only C,H → No oxygen
- Non-Polar / Hydrophillic
Common Confusion Points
- Dehydration synthesis —> forms ester or covalent bonds (ESTER BOND IS A COVALENT BOND)
- All lipids joined by covalent bonds
- For glycerol + 3 fatty acids, an ester bonds forms
- So for phospholipids or triglycerides, ester bond si formed
- All C-H bonds are still covalent bonds
Saturated Vs Unsaturated Lipids
- Saturated —> straight chains, solid at room temp (INCREASES MEMBRANE STABILITY –> HELPS AT HIGHER TEMPERATURES )
- Unsaturated —> double bonds create kinks —> liquid oils (MAKES CELL MEMBRANES MORE FLUID –> DECREASES MEMBRANE STABILITY )
- It is a liquid because these kinks prevent Vanderwaal’s forces