This section aims to provide a brief understanding of the forces in water and a more detailed understanding of different types of bonds. It is important to understand that these bonds influence structure which influences function (Examples will be shown in protein and DNA sections).
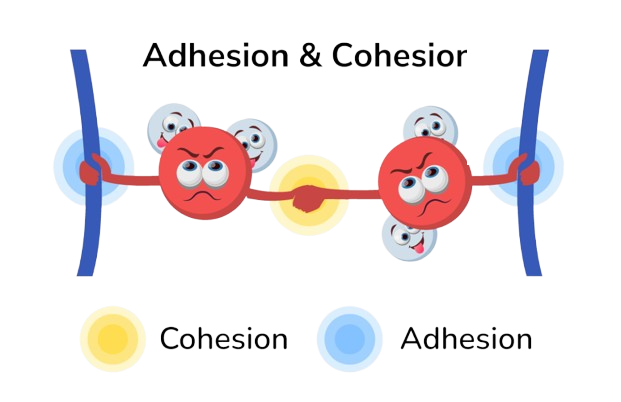
Cohesion
- Water sticks to water
- Creates surface tension (why bugs can walk on water)
- Helps move water up in plants (works with adhesion for capillary action)
Adhesion
- Water sticks to other polar surfaces
- Works with cohesion for capillary action
Capillary Action
- How water travels up plant roots/stems against gravity
- Result of cohesion + adhesion working together
Functional Groups
| Functional Group | Structure | Properties |
| Hydroxyl | R – O – H | Polar |
| Carboxyl | O || R — C — OH | Acidic |
| Amino | H | R — N | H | Basic |
| Phosphate | O || R — O — P — OH | OH | Acidic |
*Table may appear better on laptops
Tips
- Memorize the special properties of water: CHAPS (Cohesion, High specific heat, Adhesion, Polarity, Solvent)
- Hydrogen bonds are also important to the structure and function of water, and it will appear on the test